Behavioral changes to anthropogenic environments in two species of macaque
🖨︎ Printable version (PDF) LaTeX source- Submitted
- Course
- ANTH 498 Senior Capstone Seminar
- Institution
- University of Illinois at Urbana-Champaign
- Instructor
-
- Dr. Petra E. Jelinek
- Dr. Brenda M. Farnell
Abstract
Primatology is often conducted in the wild, focusing on elucidating how species live in the ecology of their native habitat. As human development has increased across the globe, more attention should be paid to how primates adapt to the anthropogenic environment. This study reviewed the published literature on Macaca fascicularis and sylvanus, comparing quantitative aspects of the species' behavior in differing degrees of anthropogenic environments. Anthropogenic habitation was expected to lead to greater consumption of human foods, smaller home ranges, longer resting times, and larger social groups. In M. fascicularis, a statistically significant relation was found between group size and habitat, but no relation was found for the other counts in either species. A call for more research into these populations' ecology and behavior is presented.
Introduction
The sciences of ecology and wildlife biology were born of a sense of exoticism, a Romanticist question of how the natural world came to be that in its climax allowed thinkers like Darwin to explore the world in pursuit of ever more novel findings and ideas (Nichols 2005). Today biologists continue to focus on the pristine, basing their models of species and ecological change on the “natural” world unmolested by human encroachment. Today, however, humans have changed the face of the planet in many cases beyond recognition from the “natural” order. Despite its frequent dismissal in ecological study, the human environment has its own ecology and unique resources that it itself should be considered from an ecological lens. An analysis of the primatology fieldwork that has heretofore been conducted in such environments is needed to understand the state of the field.
The Anthropogenic World
Human society has had a profound impact on the ecosystems of the Earth, radically changing in only a few centuries how wide swaths of the planet, terrestrial and marine, Accordingly, geologists and climate scientists have donned the current epoch the Anthropocene
(Crutzen and Stoermer 2000; Lewis and Maslin 2015). While scholars debate the exact onset of this geologic era, whether 8,000 years before present (Ruddiman 2003; Kirch 2005) or as recent as the Industrial Revolution (Crutzen 2002), it is indisputable that the majority of terrestrial life on the planet has been impacted in some way by human expansion.
Conservation of the natural environment is a laudable goal, as there is no better way to protect biodiversity and endangered species than to set aside undisturbed land (Newmark 1995; Hatfield et al. 2019). However, it cannot be denied that human settlement has drastically reduced the total land area possible for such undisturbed conservation. The anthropogenic environment is as much a part of the ecological landscape as is a coral reef, and its study can provide insight into the evolution and development of populations in an increasingly human-dominated world.
Despite this pressing need to consider anthropogenic environments, ecology research – and primatology in particular – has persistently remained far from human environments. Only between 0.4% and 6% of ecology research has been conducted in the 75% of Earth’s ice-free terrestrial surface that harbors human settlement (Collins et al. 2000; Miller and Hobbs 2002). Conservation biologists’ and ecologists’ focus on the idea of a wilderness
leaves the flora and fauna of cities little understood (Niemelä 1999), although when population censuses are taken in cities as developed as Phoenix, Arizona and Baltimore, Maryland, researchers have been surprised by the diversity present (Kloor 1999).
Anthropogenic Environments as Primate Habitats
Primatologists in particular prefer to conduct research at a small array of permanent, long-term field sites, which fails to account for the primates who are not represented (Bezanson and McNamara 2019). This model of research has of course benefits, mainly its desire to maintain relations with the local community and in its producing comparative and consistent results. Certainly the exotic field-site model has served conservationists and researchers well in the past half-century. However, understanding how the subjects of primatology research fit into the rest of the world is just as – if not more – important in the current global environment.
Most of the prevailing research of the human-nonhuman divide falls into one of two camps: that of ethnoprimatology or that of interspecies conflict management. The former primarily looks at the species divide through an ethnographic lens, in which humans and nonhuman primates together shape a social space each with shared and distinct roles (Fuentes 2012; Riley 2013). The latter consists of a diverse array of ecological scholarship aimed at reducing primate endangerment in human environments such as roadways (Lindshield 2016), analyzing primate effects on cultivated crops (Hill 2017; Hockings, Yamakoshi, and Matsuzawa 2017; McKinney, Westin, and Serio-Silva 2015), and physical conflict (Hoffman and O’Riain 2012). Recently, researchers have begun to apply focal-animal sampling techniques to primates in troops that have made their home ranges either wholly or partially inside heavily developed areas. Specifically, there is now a wealth of Macaca spp. diet and range information which can be directly compared to research on those same species in less anthropogenic environments. Urban primate populations that have undergone recent study include those of M. sylvanus in Gibraltar (Kwiatt 2017; Klegarth et al. 2017) and Béjaïa (Maibeche et al. 2015) and of M. fascicularis in Singapore (Klegarth et al. 2017).
Hypotheses
The null hypothesis is the prediction that in all surveyed characteristics, M. sylvanus and M. fascicularis individuals do not differ when in anthropogenic versus wild environments. Four alternative hypotheses are presented:
Anthropogenic-dwelling macaques exhibit a dietary shift with a bias toward human foods. The higher relative availability of human foods in such environments provides an ample source of nutrition when the foods that make up the animals’ natural diets are not obtainable.
Anthropogenic-dwelling macaques exhibit smaller ranges and shorter daily path lengths. Urban and rural human-inhabited areas present a higher density of environmental hazards, including vehicular traffic and physical conflict with people. It is predicted that in such environments, animals would travel less to avoid danger.
Time spent resting is directly correlated with consumption of human foods, while time spent feeding is indirectly correlated. The energy density of human foods is often much higher than that of foods growing in the wild, as a result of both processing and domestication. Macaques that can access such foods regularly may be able to decrease the amount of time spent feeding to reach their needed caloric intake.
Anthropogenic-dwelling macaques live in larger social groups. A lack of predation and more reliable access to food are expected to reduce a pressure toward small group sizes, allowing macaque groups in such environments to grow to larger sizes than would be expected in the wild.
Review of Literature
Collection of Data
Due to the frequency with which researchers survey urban populations of the two species, M. sylvanus and M. fascicularis were chosen as the target species for comparison. 13 studies were found to cover at least one of the two species’ behavioral patterns in anthropogenic or “wild” habitat. Data collected from each study, where present, included the environment (as urban/park, peri-urban, rural, or forest), the percentage of the group’s home range that was anthropogenic, the number (minimum, maximum, and mean) of individuals, survey method, home range (either MCP or KDE), daily path length, movement rate, diet (natural: yes, no; refuse: no, minor, extensive; sanctioned provisioned: no, yes; illicit provisioned: no, minor, extensive; and raided: no, minor, extensive, also as percentages when available), and activity budget (percent of time spent resting or feeding).
Results
Data Analysis
Data were entered into a spreadsheet and analyzed using Microsoft Excel version 1911. A list of all macaque groups identified is available in Table 1.
Hypothesis 1
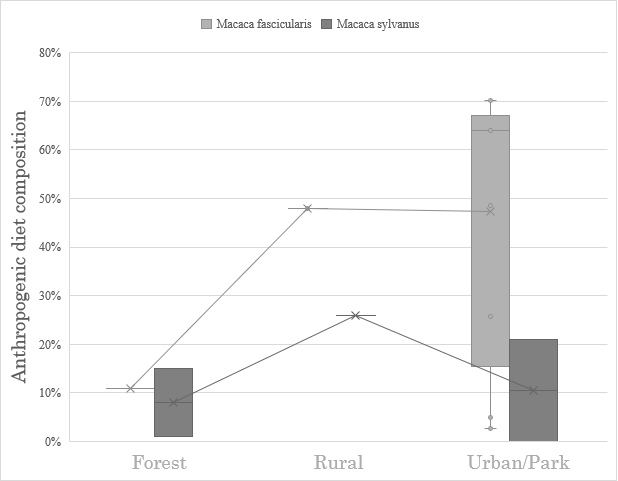
On the question of anthropogenic foods in macaque diets, very little information was found in studies of forest- or rural-dwelling animals of either species. Furthermore, there were multiple groups of urban-dwelling macaques whose anthropogenic food consumption was far lower than expected, and even lower than some forest groups (M. fascicularis min. 3%, M. sylvanus min. 0%; see Figure 1). The first hypothesis, that anthropogenic foods are correlated with anthropogenic habitation, is not accepted.
Hypothesis 2
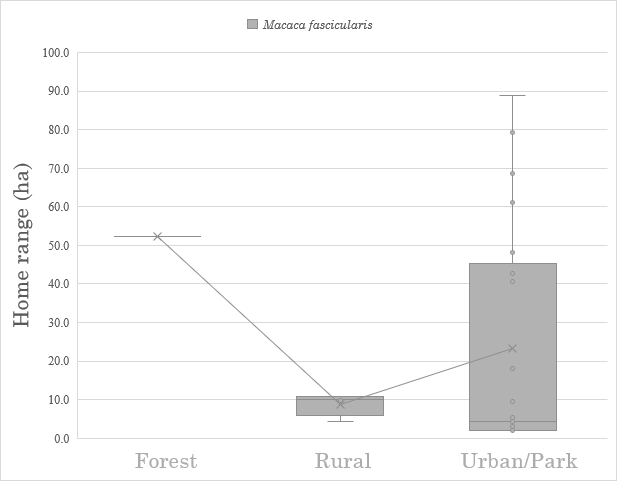
M. Sylvanus studies were excluded from home range size comparisons as they lacked enough data for the three habitat types. In M. fascicularis, while rural home ranges were overwhelmingly smaller than forest ranges, there was an extremely wide range of urban home ranges (min. 2.0 ha, mean 4.3 ha, max. 88.8 ha; see Figure 2) that overlaps the ranges for both forest and rural macaques’ home ranges. On this count there is not enough information to accept the second hypothesis.
Hypothesis 3
Few studies included both the time spent resting and the proportion of animals’ diet deriving from anthropogenic sources, with two and five in M. sylvanus and fascicularis, respectively. Making a linear model is of no use in the two-datapoint set, so only M. fascicularis was analyzed. With a positive trendline and of 0.52, there is tentative evidence of a correlation between diet and energy expenditure. Without more data, however, hypothesis 3 cannot be reliably accepted.
Hypothesis 4
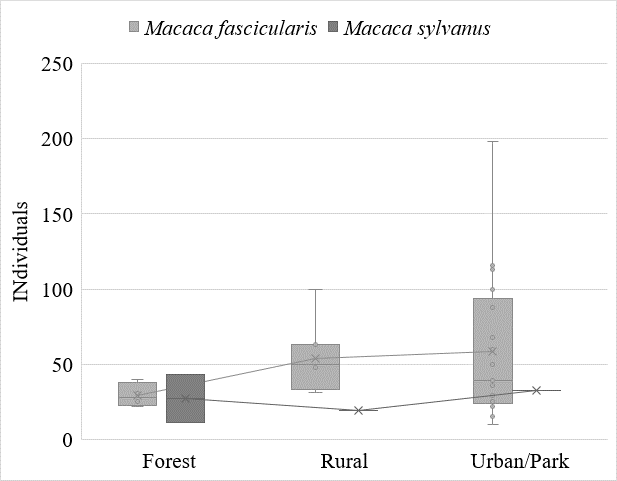
Of the 53 Macaca groups identified from the literature, 19 lacked censuses of group size. There is not enough data for a conclusive picture of M. sylvanus, but some conclusions may be drawn from the data on M. fascicularis. While forest-inhabiting groups ranged from 22 to 40 individuals in size, urban-dwelling populations existed in a much larger range of group sizes from 10 to 198 individuals, with most occurring in the 24-94 range. Albeit with large margins of error (see Figure 4), there is a statistically significant () result for group size. At least for M. fascicularis, the fourth hypothesis – that anthropogenic-dwelling macaques live in larger social groups – can be tentatively accepted.
Discussion
It was difficult to find enough primatology studies that had been conducted in anthropogenic environments, a finding reflective of Collins et al.’s (2000) finding of 0.4% of ecology research having an urban focus. A more thorough investigation of which species are most heavily focused on in such research may allow for a more comprehensive comparison. More importantly, however, it is necessary to grow the state of the primary literature so that a more comprehensive understanding of the ecologies of both cities and these species.
Overall, there is not enough published data to confirm or reject the null hypothesis, let alone any of the four alternative hypotheses proposed. There was tentative evidence for hypotheses three and four: a connection between diet and activity budget and between group size and habitat. On the former, it is necessary to perform more studies that include both anthropogenic diets and activity budget, which can allow for a larger sample size in comparisons. Furthermore, a correlation does not fully explain such a phenomenon. The energy densities of those specific foods which are eaten by the studied populations should be evaluated to ensure that the hypothesis is backed with biological significance. Few studies, even those which described anthropogenic food consumption, explained which foods the animals were eating, be they fruits or processed foods. In pursuit of the latter hypothesis, group size data on M. sylvanus would help to allow a cross-species comparison, but it remains interesting that the group sizes observed have such a wide range. It is likely that the hypothesis of lower predation allowing larger group sizes is too simple a model to explain the phenomenon of anthropogenic-dwelling macaques.
References
-
Alami, Abderrazak el, Els van Lavieren, Aboufatima Rachida, and Abderrahman Chait. 2012. “Differences in Activity Budgets and Diet Between Semiprovisioned and Wild‐feeding Groups of the Endangered Barbary Macaque (Macaca Sylvanus) in the Central High Atlas Mountains, Morocco.” American Journal of Primatology 74 (3): 210–16. doi:10.1002/ajp.21989.
-
Bezanson, Michelle, and Allison McNamara. 2019. “The What and Where of Primate Field Research May Be Failing Primate Conservation.” Evolutionary Anthropology 28 (4): 166–78. doi:10.1002/evan.21790.
-
Brotcorne, Fany. 2014. “Behavioral Ecology of Commensal Long-Tailed Macaque (Macaca Fascicularis) Populations in Bali, Indonesia: Impact of Anthropic Factors.” PhD thesis, Département de Biologie, Ecologie et Evolution; Université de Liège. https://hdl.handle.net/2268/171819.
-
Collins, James P., Ann Kinzig, Nancy B. Grimm, William F. Fagan, Diane Hope, Jianguo Wu, and Elizabeth T. Borer. 2000. “A New Urban Ecology: Modeling Human Communities as Integral Parts of Ecosystems Poses Special Problems for the Development and Testing of Ecological Theory.” American Scientist 88 (5): 416–25.
-
Crutzen, Paul J. 2002. “Geology of Mankind.” Nature 415 (23): 23. doi:10.1038/415023a.
-
Crutzen, Paul J., and Eugene F. Stoermer. 2000. “The ‘Anthropocene’.” IGBP Global Change 41: 17–18.
-
Fuentes, Agustín. 2012. “Ethnoprimatology and the Anthropology of the Human-Primate Interface.” Annual Review of Anthropology 41: 101–17. doi:10.1146/annurev-anthro-092611-145808.
-
Fuentes, Agustín, Aida L. T. Rompis, I. G. A. Arta Putra, Ni Luh Watiniasih, I. Nyoman Suartha, I. G. Soma, I. Nyoman Wandia, I. D. K. Harya Putra, Rebecca Stephenson, and Wayan Selamet. 2011. “Macaque Behavior at the Human-Monkey Interface: The Activity and Demography of Semi-Free-Ranging Macaca Fascicularis at Padangtegal, Bali, Indonesia.” In Monkeys on the Edge: Ecology and Management of Long-Tailed Macaques and Their Interface with Humans, edited by Michael D. Gumert, Agustín Fuentes, and Lisa Jones-Engel, 159–82. Cambridge University Press.
-
Hatfield, Jack H., Jos Barlow, Carlos A. Joly, Alexander C. Lees, Ceslo Henrique de Freitas Parruco, Joseph A. Tobias, C. David L. Orme, and Cristina Banks-Leite. 2019. “Mediation of Area and Edge Effects in Forest Fragments by Adjacent Land Use.” Conservation Biology Forthcoming. doi:10.1111/cobi.13390.
-
Hill, Catherine M. 2017. “Primate Crop Feeding Behavior, Crop Protection, and Conservation.” International Journal of Primatology 38 (2): 385–400. doi:10.1007/s10764-017-9951-3.
-
Hockings, Kimberley J., Gen Yamakoshi, and Tetsuro Matsuzawa. 2017. “Dispersal of a Human-Cultivated Crop by Wild Chimpanzees (Pan Troglodytes Verus) in a Forest–Farm Matrix.” International Journal of Primatology 38 (2): 172–93. doi:10.1007/s10764-016-9924-y.
-
Hoffman, Tali S., and M. Justin O’Riain. 2012. “Monkey Management: Using Spatial Ecology to Understand the Extent and Severity of Human-Baboon Conflict in the Cape Peninsula, South Africa.” Ecology and Society 17 (3): 13. doi:10.5751/ES-04882-170313.
-
Ilham, Kurnia, Rizaldi, Jabang Nurdin, and Yamato Tsuji. 2017. “Status of Urban Populations of the Long-Tailed Macaque (Macaca Fascicularis) in West Sumatra, Indonesia.” Primates 58 (2): 295–305. doi:10.1007/s10329-016-0588-1.
-
Kirch, Patrick V. 2005. “Archaeology and Global Change: The Holocene Record.” Annual Review of Environment and Resources 30: 409–40. doi:10.1146/annurev.energy.29.102403.140700.
-
Klegarth, Amy R., Hope Hollocher, Lisa Jones-Engel, Eric Shaw, Benjamin P. Y.-H. Lee, Tessa Feeney, Damian Holmes, Dale Laguea, and Agustín Fuentes. 2017. “Urban Primate Ranging Patterns: GPS‐collar Deployments for Macaca Fascicularis and M. Sylvanus.” American Journal of Primatology 79 (5): e22633. doi:10.1002/ajp.22633.
-
Kloor, Keith. 1999. “A Surprising Tale of Life in the City.” Science 286 (5440): 663. doi:10.1126/science.286.5440.663.
-
Kwiatt, Anne. 2017. “Food, Feeding, and Foraging: Using Stable Isotope Analysis as a Methodology in the Study of Urban Primate Dietary Patterns.” In Ethnoprimatology: A Practical Guide to Research at the Human-Nonhuman Primate Interface, edited by Kerry M. Dore, Erin P. Riley, and Agustín Fuentes, 56–69. Cambridge: Cambridge University Press.
-
Lewis, Simon L., and Mark A. Maslin. 2015. “Defining the Anthropocene.” Nature 519: 171–80. doi:10.1038/nature14258.
-
Lindshield, Stacy M. 2016. “Protecting Nonhuman Primates in Peri-Urban Environments: A Case Study of Neotropical Monkeys, Corridor Ecology, and Coastal Economy in the Caribe Sur of Costa Rica.” In Ethnoprimatology, edited by Michel T. Waller, 351–69. Springer Nature. doi:10.1007/978-3-319-30469-4_19.
-
Maibeche, Yasmina, Aissa Moali, Nassima Yahi, and Nelly Menard. 2015. “Is Diet Flexibility an Adaptive Life Trait for Relictual and Peri-Urban Populations of the Endangered Primate Macaca Sylvanus?” PLOS One 10 (2): e0118596. doi:10.1371/journal.pone.0118596.
-
McKinney, Tracie, Jessica L. Westin, and Juan Carlos Serio-Silva. 2015. “Anthropogenic Habitat Modification, Tourist Interactions and Crop-Raiding in Howler Monkeys.” In Howler Monkeys, edited by Martín M. Kowalewski, Paul A. Garber, Liliana Cortés-Ortiz, Bernardo Urbani, and Dionisios Youlatos, 281–311. New York: Springer Nature. doi:10.1007/978-1-4939-1960-4_11.
-
Md-Zain, Badrul Munir, Mohamed Reza Tarmizi, and Mastura Mohd-Zaki. 2011. “Campus Monkeys of Universiti Kebangsaan Malaysia: Nuisance Problems and Students’ Perceptions.” In Monkeys on the Edge: Ecology and Management of Long-Tailed Macaques and Their Interface with Humans, edited by Michael D. Gumert, Agustín Fuentes, and Lisa Jones-Engel, 101–17. Cambridge University Press.
-
Miller, James R., and Richard J. Hobbs. 2002. “Conservation Where People Live and Work.” Conservation Biology 16 (2): 330–37. doi:10.1046/j.1523-1739.2002.00420.x.
-
Newmark, William D. 1995. “Extinction of Mammal Populations in Western North American National Parks.” Conservation Biology 9 (3): 512–26. doi:10.1046/j.1523-1739.1995.09030512.x.
-
Nichols, Ashton. 2005. “Roaring Alligators and Burning Tygers: Poetry and Science from William Bartram to Charles Darwin.” Proceedings of the American Philosophical Society 149 (3): 304–15.
-
Niemelä, Jari. 1999. “Ecology and Urban Planning.” Biodiversity & Conservation 8 (1): 119–31. doi:10.1023/A:1008817325994.
-
Noordwijk, Maria A. van, and Carcl P. van Schaik. 1999. “The Effects of Dominance Rank and Group Size on Female Lifetime Reproductive Success in Wild Long-Tailed Macaques, Macaca Fascicularis.” Primates 40 (1): 105–30. doi:10.1007/BF02557705.
-
Riley, Erin P. 2013. “Contemporary Primatology in Anthropology: Beyond the Epistemological Abyss.” American Anthropologist 115 (3): 411–22. doi:10.1111/aman.12025.
-
Ruddiman, William F. 2003. “The Anthropogenic Greenhouse Era Began Thousands of Years Ago.” Climatic Change 61 (3): 261–93. doi:10.1023/B:CLIM.0000004577.17928.fa.
-
Schurr, Mark R., Agustín Fuentes, Ellen Luecke, John Cortes, and Eric Shaw. 2012. “Intergroup Variation in Stable Isotope Ratios Reflects Anthropogenic Impact on the Barbary Macaques (Macaca Sylvanus) of Gibraltar.” Primates 53 (1): 31–40. doi:10.1007/s10329-011-0268-0.
-
Sha, John Chih Mun, and Goro Hanya. 2013. “Diet, Activity, Habitat Use, and Ranging of Two Neighboring Groups of Food‐enhanced Long‐tailed Macaques (Macaca Fascicularis).” American Journal of Primatology 75 (6): 581–92. doi:10.1002/ajp.22137.
-
Wong, Che-lok. 1994. “Studies on the Feral Macaques of Hong Kong.” Master’s thesis, Hong Kong: Hong Kong University of Science and Technology.
| Food | Time spent | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Source | Species | Location | Name | Habitat | Urban1 | Individuals | KDE (ha)2 | DPL (km)3 | Garbage | Provisioned4 | Raided | Total | Resting | Feeding |
| Maibeche et al. (2015) | sylvanus | Béjaïa | Les Oliviers | Urban | 33 | No | Moderate | No | 0.21 | |||||
| Cap Carbon | Forest | 43 | No | Minor | No | 0.15 | ||||||||
| Klegarth et al. (2017) | fascicularis | Singapore | SG-BB | Urban | 0.58 | 35 | 1.37 | Moderate | Minor | |||||
| SG-BNTR 01 | Urban | 0.05 | 34.70 | 1.56 | Minor | No | ||||||||
| SG-BNTR 02 | Urban | 0.19 | 23.8 | 0.72 | Moderate | Minor | ||||||||
| SG-BTRR | Urban | 0 | 42.7 | 0.95 | Minor | No | ||||||||
| SG-MNCF | Urban | 0.11 | 40.80 | 2.16 | Moderate | Minor | ||||||||
| SG-RR | Urban | 0 | 54.7 | 1.11 | No | Minor | ||||||||
| SG-WW | Urban | 0 | 25.9 | 1.53 | Minor | Moderate | ||||||||
| sylvanus | Gibraltar | GIB-AD | Urban | 0.36 | 12.4 | 1.48 | Moderate | Extensive | ||||||
| GIB-CC | Urban | 0 | 9.5 | 1.4 | Minor | Extensive | ||||||||
| GIB-LBI | Urban | 0 | 10.8 | 1.09 | Moderate | No | ||||||||
| GIB-MH | Urban | 0.27 | 83.3 | 3.46 | Moderate | Moderate | ||||||||
| GIB-PPA | Urban | 0 | 13.7 | 1.21 | Minor | Extensive | ||||||||
| GIB-RAW | Urban | 0 | 20.10 | 1.6 | Moderate | Extensive | ||||||||
| Sha and Hanya (2013) | fascicularis | Singapore | High | Urban | 26 | 9.5 | 1.48 | Moderate | Moderate | 0.49 | 0.14 | 0.49 | ||
| Low | Urban | 22 | 18.2 | 1.8 | Minor | Moderate | 0.26 | 0.23 | 0.46 | |||||
| Ilham et al. (2017) | fascicularis | West Sumatra | A | Urban | 36 | 2 | Moderate | Moderate | 0.7 | |||||
| B | Urban | 28 | 2 | Moderate | Moderate | 0.7 | ||||||||
| C | Urban | 68 | 2 | Moderate | Moderate | 0.7 | ||||||||
| X | Urban | 15 | 2 | Minor | Minor | 0.05 | ||||||||
| G | Urban | 10 | 2 | Minor | Minor | 0.03 | ||||||||
| P | Urban | 15 | 2 | Minor | Minor | 0.03 | ||||||||
| Wong (1994) | fascicularis | Kowloon | E | Urban | 34 | |||||||||
| Alami et al. (2012) | sylvanus | Ozoud Falls | Semi-provisioned | Rural | 19 | 0.26 | 0.17 | 0.28 | ||||||
| Wild-feeding | Forest | 11 | 0.01 | 0.14 | 0.26 | |||||||||
| Fuentes et al. (2011) | fascicularis | Bali | 1 | Rural | 31 | 7.2 | No | Extensive | 0.23 | |||||
| 2 | Rural | 100 | 11 | No | Extensive | 0.23 | ||||||||
| 3 | Rural | 63 | 17 | No | Extensive | 0.23 | ||||||||
| Md-Zain, Tarmizi, and Mohd-Zaki (2011) | fascicularis | Universiti Kebangsaan Malaysia | LF | Urban | 60 | Moderate | Minor | |||||||
| KBH | Urban | 39 | Moderate | Minor | ||||||||||
| KIY | Urban | 37 | Moderate | Minor | ||||||||||
| KAB | Urban | 23 | Moderate | Minor | ||||||||||
| KRK | Urban | 52 | Moderate | Minor | ||||||||||
| KIZ | Urban | 30 | Moderate | Minor | ||||||||||
| KKM | Urban | 50 | Moderate | Minor | ||||||||||
| PBP | Urban | 100 | Moderate | Minor | ||||||||||
| Schurr et al. (2012) | sylvanus | Gibraltar | AD | Urban | 0.77 | |||||||||
| PPA | Urban | 0.76 | ||||||||||||
| MH | Urban | 0.5 | ||||||||||||
| Brotcorne (2014) | fascicularis | Bali | TNBBg | Forest | 0.14 | 25 | 32.5 | Moderate | No | Minor | 0.11 | 0.19 | 0.34 | |
| Nez | Rural | 0.52 | 48 | 6.6 | Minor | No | Moderate | 0.48 | 0.23 | 0.27 | ||||
| Tear | Rural | 0.52 | 33 | 5.9 | Minor | No | Moderate | 0.48 | 0.23 | 0.27 | ||||
| Temple2 | Rural | 0.52 | 50 | 2 | Minor | No | Moderate | 0.48 | 0.23 | 0.27 | ||||
| Scarface | Rural | 0.52 | 50 | 9.5 | Minor | No | Moderate | 0.48 | 0.23 | 0.27 | ||||
| Cemetery | Urban | 0.74 | 88 | 1.7 | Minor | No | Extensive | 0.64 | 0.32 | 0.21 | ||||
| East | Urban | 0.74 | 113 | 4.10 | Minor | No | Extensive | 0.64 | 0.32 | 0.21 | ||||
| Michelin | Urban | 0.74 | 116 | 2.9 | Minor | No | Extensive | 0.64 | 0.32 | 0.21 | ||||
| Central | Urban | 0.74 | 102 | 4.40 | Minor | No | Extensive | 0.64 | 0.32 | 0.21 | ||||
| Temple | Urban | 0.74 | 198 | 3.4 | Minor | No | Extensive | 0.64 | 0.32 | 0.21 | ||||
| Noordwijk and Schaik (1999) | fascicularis | North Sumatra | A | Forest | 0 | 22 | ||||||||
| H | Forest | 0 | 40 | |||||||||||
| K | Forest | 0 | 31 | |||||||||||